Two projects are currently active in Ballard's research group.
1. A Laboratory Sequence Linking Organic Chemistry II and Biochemistry Laboratory Courses
This project is conducted in collaboration with Scott Witherow, a faculty member in the department whose expertise includes biochemistry and biochemistry education.
The objective of this experiment is to help break down the perceived silos between these two classes by designing and implementing a sequence that spans them. Current Biochemistry Laboratory (CHE 320L) students focus on a semester-long sequence that includes expression, purification and kinetic studies of the enzyme alkaline phosphatase.
The kinetic studies include the use of inhibitors to study their effects. The extension offered by this project is to add an experiment to Organic Chemistry II Laboratory (CHE 235L) that prepares potential inhibitors of the enzyme by derivatizing the amino acid phenylalanine. It is envisioned that the same student could prepare a derivative in CHE 235L during one semester and study it as a potential enzyme inhibitor in CHE 320L during the following semester.
Ballard's group is preparing several categories of derivatives to determine the optimal one for the project. This project might serve as an introduction to research in Ballard's group.
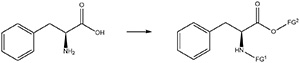
Fig 1:Derivatization of phenylalanine to form a potential inhibitor.
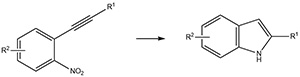
Fig 2:Transformation of a 2-nitroarylacetylene into a 2-substituted indole.
2. Preparation of a Modified Indole Structure Using Two Chemical Reactions in One Flask
This project focuses on preparation of modified forms of indole. The indole core is present in many biologically active molecules, including the amino acid tryptophan, the neurotransmitter serotonin, the hormone melatonin, and the psychedelic drug psilocybin. Because of their interesting biological effects, several methods have been discovered for synthesizing compounds with the indole core.3 However, the investigation of new methods is still warranted.
This project aims to probe the preparation of one category of indole-based compounds (2-substituted indoles) using a cascade approach of two reactions occurring sequentially in the same reaction vessel. Similar approaches are known using expensive and/or toxic catalysts;4 this approach is different because it will probe the effects of inexpensive chemicals of low toxicity, such as those based on iron, zinc, or boron. This project lies in the area of green chemistry because it involves the use of safer catalysts and/or safer solvents.
This project is appropriate for students interested in a multi-semester project in discovery of synthetic organic methods.
Works Cited
1. Witherow, D.S. "A Ten-Week Biochemistry Lab Project Studying Wild-Type and Mutant Bacterial Alkaline Phosphatase," Biochem. Mol. Biol. Educ. 2016, 44, 555-564.
2. Dean, R. L. "Kinetic Studies with Alkaline Phosphatase in the Presence and Absence of Inhibitors and Divalent Cations," Biochem. Mol. Biol. Educ. 2002, 30, 401-407.
3. Lead reference: Sundberg, R. J., Ed. Indoles, 1996.
4. Lead reference: Yamane, Y.; Liu, X.; Hamasaki, A.; Ishida, T.; Haruta, M.; Yokoyama, T.; Tokyunaga, M. "One-Pot Synthesis of Indoles and Aniline Derivatives from Nitroarenes Under Hydrogenation Condition with Supported Gold Nanoparticles," Org. Lett. 2009, 11, 5162-5165.