
Eric J. Werner
Professor of Chemistry
- 2002 B.S. in Chemistry with Highest Honors, University of Florida; Mentor: Professor Michael J. Scott
- 2007 Ph.D. in Chemistry, University of California, Berkeley; Mentor: Professor Kenneth N. Raymond
Research Interests
Research in our lab focuses on the synthesis of new molecules capable of binding specific lanthanide or actinide metal ions for a variety of biomedical and environmental applications. Novel f-element chelators are being prepared for applications ranging from luminescent sensors to extractants for improved lanthanide/actinide separation technologies. For example, complexes of select lanthanide ions (e.g. Eu3+ and Tb3+) are currently being evaluated with regard to their luminescent properties to enable imaging or sensing of biologically relevant targets. The relatively long luminescence decay lifetimes, sharp emission bands and large Stokes shifts of the sensitized emission make these ions ideal as probes under biological conditions. In addition to these sensor studies, current collaborative extensions of the work involve the encapsulation of the small molecule chelates within silica-based nanoparticles. The combination of the unique photophysical properties of the lanthanides with the well-known chemistry and diverse applicability of silica nanoparticles may yield highly emissive materials for use in a variety of areas including biomedical imaging.
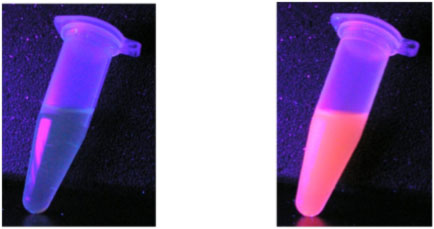
Solution of a Eu complex excited by UV light (left) and the same complex encapsulated by silica nanoparticles illuminated under identical conditions (right).
With support from the National Science Foundation (NSF grant award no. 2102381), current directions within the lab have also focused on the development of selective extractants of rare earth elements and, in some cases, actinides. Due to the large volumes of nuclear waste materials already present and the need for alternative energy sources which may include nuclear, improved methods for waste remediation due to safety and environmental concerns are desirable. The increasing use of lanthanide metals across many areas of modern technology and alternative energy applications also creates a need for acquiring these valuable metals more efficiently. The development of improved f-element extraction agents would enable metal mining with less environmental impact as well as allow for rare earth element recycling from electronic waste. Fundamental to this challenge is the design of molecules with enhanced metal extraction capability, inspiring further chelator development and f-element coordination chemistry research in our group.

Undergraduate student Tavya Benjamin synthesizing a new macrocyclic Eu3+ complex for luminescent sensor applications.
Chemistry is a hands-on, experimental science. The time students spend in the classroom learning about chemistry is essential in providing the necessary theory and background; it is in the research lab where students come full circle by putting this knowledge into action. In my lab, students are given the opportunity (and the great challenge!) of conducting their own original research projects, applying what they have learned through their coursework as well as acquiring new knowledge and skills in a relevant area of modern chemical research. In carrying out the work noted above, students are involved in every aspect of the project: from the design of new molecules or novel assays to the final presentation and publication of the more promising results. Also, due to the interdisciplinary nature of the work, students are exposed to a variety of areas including inorganic and organic synthesis in preparing novel lanthanide complexes, analytical chemistry in performing various assays and through characterization of all new molecules synthesized, physical chemistry in rationalizing the magnetic and photophysical properties of the imaging agents and biomedical research in exploring potential applications. Beyond learning basic synthetic techniques, students routinely have full access to a number of sophisticated instruments on campus including NMR (300 MHz), FTIR, UV-Vis and fluorescence spectrometers.
If you are interested in learning more or would like to discuss research opportunities in my lab, please contact me at ewerner@ut.edu or stop by my office.
(Undergraduate student co-authors' names are in bold type.)
Werner, E. J.; Biros, S. M. “Supramolecular Ligands for the Extraction of Lanthanide and Actinide Ions.” Org. Chem. Front. 2019, 6, 2067–2094.
Patterson, M. G.; Mulville, A. K.; Connor, E. K.; Henry, A. T.; Tissue, K.; Biros, S. M.; Werner, E. J. "Lanthanide Extraction Selectivity of a Tripodal Carbamoylmethylphosphine Oxide Ligand System.” Dalton Trans. 2018, 47, 14318–14326.
Leach, E. G.; Shady, J. R.; Boyden, A. C.; Emig, A.; Henry, A. T.; Connor, E. K.; Staples, R. J.; Schaertel, S.; Werner, E. J.; Biros, S. M. “X-ray Crystallographic, Luminescence and NMR Studies of Phenacyldiphenylphosphine Oxide with the Ln(III (3)) ions Sm, Eu, Gd, Tb and Dy.” Dalton Trans. 2017, 46, 15458–15469.
Coburn, K. M.; Hardy, D. A.; Patterson, M. G.; McGraw, S. N.; Peruzzi, M. T.; Boucher, F.; Beelen, B.; Sartain, H. T.; Neils, T.; Lawrence, C. L.; Staples, R. J.; Werner, E. J.; Biros, S. M. “ f-Element Coordination and Extraction Selectivity of a Carbamoylmethylphosphine Oxide Ligand Based on a Tripodal Phosphine Oxide Scaffold.” Inorg. Chim. Acta, 2016, 449, 96–106.
Duncan, A. K.; McBride, C. N.; Benjamin, T. G. R.; Madsen, M. P.; Bowers, K. T.; de Bettencourt-Dias, A.; Werner, E. J. “Tuning the Structural and Lanthanide Luminescence Properties of Macrocyclic Tetraiminodiphenolate Europium(III (3)) Complexes.” Polyhedron, 2016, 114, 451–458, Invited paper for special issue: “Undergraduate Research in Inorganic Chemistry.”
Werner, E. J.; Benjamin, T. G. R. “Molecular Gadolinium Complexes for Magnetic Resonance Imaging.” McGraw-Hill Yearbook of Science & Technology 2014; McGraw-Hill: New York, 2014.
Makhinson, B. Duncan, A. K.; Elam, A. R.; de Bettencourt-Dias, A.; Medley, C. D.; Smith, J. E.; Werner, E. J. “Turning on Lanthanide Luminescence via Nanoencapsulation.” Inorg. Chem. 2013, 52, 6311–6318.
(Undergraduate student co-authors' names are in bold type. Presenting author underlined.)
Lake, B. J.; Werner, E. J. "Effect of Scaffold Variation of Tripodal Carbamoylmethylphosphine Oxide Ligands for Lanthanide Extraction." Spring ACS National Meeting, San Diego, CA, March 2022.
Becht, P.; Lake, B. J.; Larrinaga, W. B.; Biros, S. M.; Werner, E. J. "Alteration of Rare Earth Extraction Protocol Conditions: Toward Enhanced Lanthanide Metal Separations." Spring ACS National Meeting, San Diego, CA, March 2022.
Larrinaga, W.; Martinez, A.; Werner, E. J. "Coordination Chemistry, Luminescence, and f-Element Extraction Selectivity of a Tripodal Carbamoylmethylphosphine Oxide-Based Ligand." 71st Southeastern Regional Meeting of the ACS, Savannah, GA, October 2019.
Werner, E. J.; Polzin, S. M.; Felix, K. H. “Lanthanide luminescence and anion binding properties of tripodal iminopyridine complexes.” 255th ACS National Meeting, New Orleans, LA, March 2018.
Werner, E. J. “Development of CMPO-Based Ligands for Selective Lanthanide Extraction.” 28th Rare Earth Research Conference, Iowa State University, Ames, IA, June 2017, Invited talk.
Werner, E. J.; Botta, M.; Johnson, K. R.; Madsen, M. P. “Linking Fundamental Undergraduate f-Element Research to Real-World Applications: Development of Pyridine/Phosphonate Based MRI Contrast Agents.” 247th ACS National Meeting, Dallas, TX, March 2014, Invited talk.

