|
John Struss Education:
|
Research Interests
Hypervalent Halogen Amine Complexes
Over the past few decades, hypervalent halogen complexes such as the Dess-Martin (DMP) and IBX reagents have been used in a wide variety of applications. These include the conversion of alcohols, imines, thioketals and benzylic methylenes (Ph-CH2-R) to ketones, aldehydes, amines, imines, phenols to quinones, and the synthesis of δ-lactams and cyclic urethanes. Although such reagents are desirable because of their versatility, they are somewhat expensive (approximately $300/mol), shock-sensitive, and suffer from an unfavorable atom economy in most applications. In an effort to find a more attractive alternative to such reagents, we are exploring the uses of hypervalent bromine (I) amine complexes first developed by Blair and co-workers.
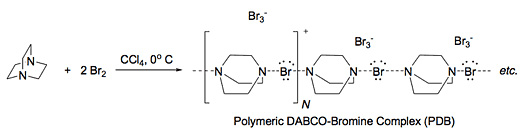
Quinuclidine and related compounds react with molecular bromine to give hypervalent complexes such as bis-quinuclidine bromine (I) bromide (BQBB) and polymeric DABCO-bromine complex (PDB; Fig. 2). It has already been established that such complexes are mild oxidants converting secondary and primary alcohols to the corresponding ketone and aldehyde. These reagents are chemoselective for the oxidation of secondary hydroxyls over primary.
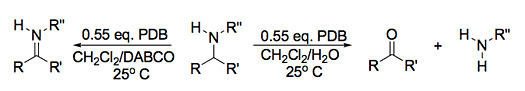
Recently, we have discovered that these reagents, like the IBX and DMP reagents, also exhibit a variety of reactivity and are a shelf-ready source of electrophilic and free-radical bromine. For example, PDB is capable of converting amines to imines, or ketones and/or aldehydes, depending on the reaction conditions (Scheme 1). This complex is also a high-yielding para-selective brominating reagent for aromatic systems with strong electron-donating substituents (Scheme 2). It has also been observed that PDB brominates aromatic hydrocarbons such as toluene, ethylbenzene and cumene are converted to benzyl bromide, (1-bromoethyl) benzene, and 2-bromo-2-phenylpropane, the latter of which eliminates rapidly to α-methyl styrene under biphasic CH2Cl2/H2O reaction conditions. We hope to carefully study the scope and limitations of these synthetic routes to provide an alternative to the somewhat unattractive IBX and DMP reagents.
The Asymmetric Baylis-Hillman Reaction – Cyclodextrin as a Chiral Additive and Catalyst Support
The field of organocatalysis has grown dramatically over recent years. The notion of mimicking enzymatic processes using relatively small and inexpensive compounds is certainly an attractive one. By far, nucleophilic, nitrogen-based compounds are the most popular. Organocatalysts such as derivatives of proline, quinuclidine, indole, imidazole, dimethyl aminopyridine (DMAP), and various oligopeptides have been used in a variety of applications including aldol, Mannich, Michael, cycloaddition, cyclopropanation, Stetter, Baylis-Hillman, epoxidation, and acyl transfer reactions.
The Baylis-Hillman reaction (Scheme 3) is of particular interest because it possesses several attractive features found in the most useful organic transformations. First, Baylis-Hillman adducts contain several functional groups and a new chiral center; a hydroxyl group bound to a chiral carbinol and an alkene in conjugation with an electron-withdrawing group, typically carbonyl, imidyl, thionyl or nitrile. Baylis-Hillman adducts are, therefore, useful synthons for the synthesis of more complex compounds. Second, the most widely used catalyst is typically a derivative of quinuclidine; a bicyclic tertiary amine found in the cinchona class of natural products and other chiral reagents used in asymmetric catalytic processes (Fig. 3).
Lastly, the reaction generally features a favorable atom economy. This reaction has its drawbacks, however. Most notably, the reaction requires long reaction times that are typically on the order of hours or days. Slow reaction rates may be overcome by running the reaction at high pressures and/or concentrations, using highly electron-deficient alkenes or aldehydes, or the use of hydrogen bonding solvents or additives. Increased temperatures also enhance the rate although, interestingly, there is at least one account of an increase in reaction rate as temperature decreases.
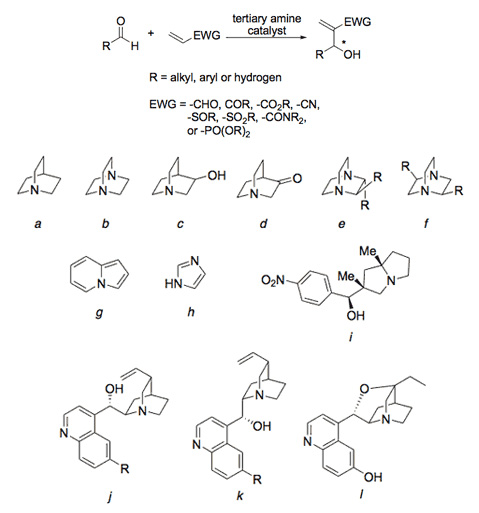
Figure 3. Baylis-Hillman catalysts: a) quinuclidine, b) 1,4-diazabicyclo[2.2.2]octane (DABCO), c) 3-hydroquinuclidine, d) 3-quinuclidone, e) C-2 symmetric 2,3-diphenyl DABCO (-R = -Ph) or
2,3-dialkoxy DABCO (-R = -OR), f) C-2 symmetric 2,5-dibenzyl DABCO (-R = -CH2Ph), g) indolizine, h) imidazole, i) a chiral derivative, j) quinidine (-R = -OMe) or cinchonine (-R = -H),
k) quinine (-R = -OMe) or cinchonidine (-R = -H), and l) β-isocupreidine.
The most widely accepted mechanism is shown in Scheme 4 (e. g. acrylic ester and benzaldehyde). In the first step, the nucleophilic catalyst adds in a Michael fashion to an activated (electron-poor) alkene to give a zwitter ionic enolate. The newly generated enolate is then available to add in an aldol fashion to electrophiles such as aldehydes and ketones. Subsequent proton transfer and catalyst regeneration steps complete the cycle.
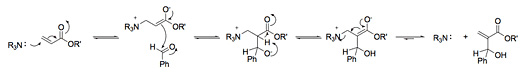
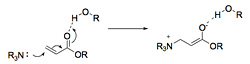
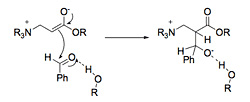
Hydrogen bonding has been shown to enhance the rate. Two to 10 fold increases have been reported in the presence of hydrogen bond donors. Hydrogen bonding solvents and additives may enhance the rate by 1) activating the alkene further, and thus enhancing the addition of the nucleophilic catalyst and/or stabilizing the resulting enolate (Scheme 5) or 2) activating the electrophile (aldehyde) making it more reactive in the aldol step and stabilizing the alkoxide intermediate (Scheme 6), or 3) a combination of these effects. Several investigators have made compelling arguments supporting each rationale, but the effect of hydrogen bonding is still unclear.
Electron demand also has an effect on the reaction rate. Ethyl acrylate (-EWG = -CO2Et; Scheme 3), for example, reacts with benzaldehyde slower than 2,2,2-trifluoroethyl acrylate (EWG = -CO2CH2CF3).
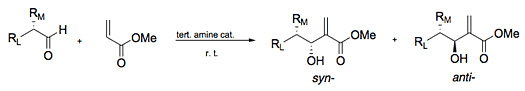
Over the past 15 years, efforts to develop an asymmetric Baylis-Hillman reaction have been moderately successful. The majority of these endeavors have taken advantage of two of the three major strategies for stereocontrol: substrate control and asymmetric catalysis. Regarding substrate controlled processes, the reaction is moderately Felkin-Ahn selective (Scheme 7) usually giving product ratios ranging from 1.2 : 1 to 7 : 1 favoring the Felkin (syn-) product. Chiral auxiliary approaches have demonstrated varying degrees of stereoselectivity (e. g. eqs. 1 - 3) with auxiliary bound acrylates and acrylamides. However, in order to obtain Baylis-Hillman adducts, such strategies require two additional steps, the attachment and removal the auxiliary or an additional modification, and are therefore not favorable in an atom economy sense.
Asymmetric catalysts, like C-2 symmetric catalysts and the cinchona alkaloids (Fig. 3, compounds e, f, j, and k), have exhibited low to moderate stereoselectivities (0 – 47% ee) with acrylonitrile and methyl vinyl ketone. Improvements were made with a variety of alkenes and electrophiles using a pyrrolizidine derivative (Fig. 3, compound i) giving stereoselectivities of 30 – 75% ee. High selectivities (91 – 99% ee) have been observed while using β-isocupreidine (Fig. 3, compound l) at -55 oC with the highly reactive acrylate 1,1,1,3,3,3-trifluoroisopropyl acrylate (-R’ = -CH(CF3)2, Scheme 2).
At low temperatures, high selectivities are observed only for the activated acrylate and, like chiral auxiliary approaches, the removal of the trifluoroisopropyl group from the adduct is often required for the synthesis of more complex compounds. The rationales for high stereoselectivities when using both β-isocupreidine and the pyrrolizidine derivative address the idea that both catalysts have hydrogen bond donors and act as bifunctional nucleophilic/hydrogen bond donating organocatalysts.
We hope to develop a new method for an asymmetric Baylis-Hillman reaction using DABCO as a catalyst and cyclodextrin or a cyclodextrin derivative as a co-catalyst/chiral additive. Cyclodextrins are macrocyclic oligomers composed of glucopyranose subunits (cyclic oligosaccharides) and are commercially available as α-, β-, and γ- cyclodextrins which contain six, seven, and eight subunits respectively. These compounds are also available commercially as a variety of derivatives (Fig. 5) and are widely used in chiral chromatographic applications.
There are a few recent examples of their use as chiral supports for transition metals and other reagents for asymmetric reductions and cyclopropanation reactions. In much of the cyclodextrin literature, the structure is often described as taking on a conical “lampshade” shape with the primary hydroxyls oriented near the narrow opening of the “lampshade” while the secondary hydroxyls are found on the opposite side (Fig. 6). Cyclodextrins and their derivatives have been described as “artificial enzymes” and are known to interact with organic compounds in a host-guest manner such that their hydrophobic cavities tend to encapsulate and release nonpolar compounds.
It has also been suggested that, for compounds such as benzaldehyde, the aryl portion of the aldehyde finds itself encapsulated within the hydrophobic cavity while the carbonyl is held outside the cavity because of hydrogen bonding. For Baylis-Hillman applications, restricted mobility of the electrophile and/or the alkene within a bulky chiral template may lead to high stereoselectivities. Furthermore, hydrogen bond donation of cyclodextrin may also enhance the reaction rate.
We have conducted a preliminary study using methyl acrylate, a variety of aldehydes, DABCO and α-cyclodextrin as an additive/co-catalyst. Our data show that yields are slightly higher when cyclodextrin is present compared to when it is absent over the same time period. We have yet to determine the effect cyclodextrin might have on the stereoselectivity of this reaction. Not surprisingly, we have discovered that α-cyclodextrin is insoluble in a number of conventional solvents.
However, in many small molecular weight aldehydes (used in a 13 to 14 fold excess with respect to acrylate), cyclodextrin is soluble at room temperature and, when no solvent is used, we have observed that the rate enhancement is even more pronounced compared to reactions in which solvents such as DMSO, DMF, THF or CH2Cl2 are used. This is particularly encouraging from a “green chemistry” standpoint. We hope to continue this study using other large cyclic oligosaccharides as chiral additives/co-catalysts.
Expanding on the theme of bifunctional organocatalysts, it is hoped that this project might be developed further by binding the nucleophilic catalyst directly to the cyclodextrin macrocycle. Luckily, there is a substantial amount of literature addressing the manipulation and functionalization of cyclodextrins. To bind quinuclidine and other nucleophilic catalysts to the primary face of cyclodextrins, we will follow Yi’s well-established protocol for primary face monoalkylation which involves protection of the primary face, exhaustive methylation of the secondary face, deprotection of the primary face and finally monotosylation or mesylation of the primary face. Yi has shown that the tosylate is easily displaced by SN2 reaction using alkoxides as nucleophiles. In our case we will use the alkoxide of 3-hydroxyquinuclide (compound c, Fig. 3).
Similar strategies have also been used to modify the secondary face of cyclodextrins. There are also examples in the literature of using imidazole (compound h, Fig. 3) as a nucleophilic reagent to displace the tosylate ultimately giving imidazole modified cyclodextrins. In short, we hope to generate a cyclodextrin-supported quinuclidine and imidazole catalysts for use in the Baylis-Hillman reaction.